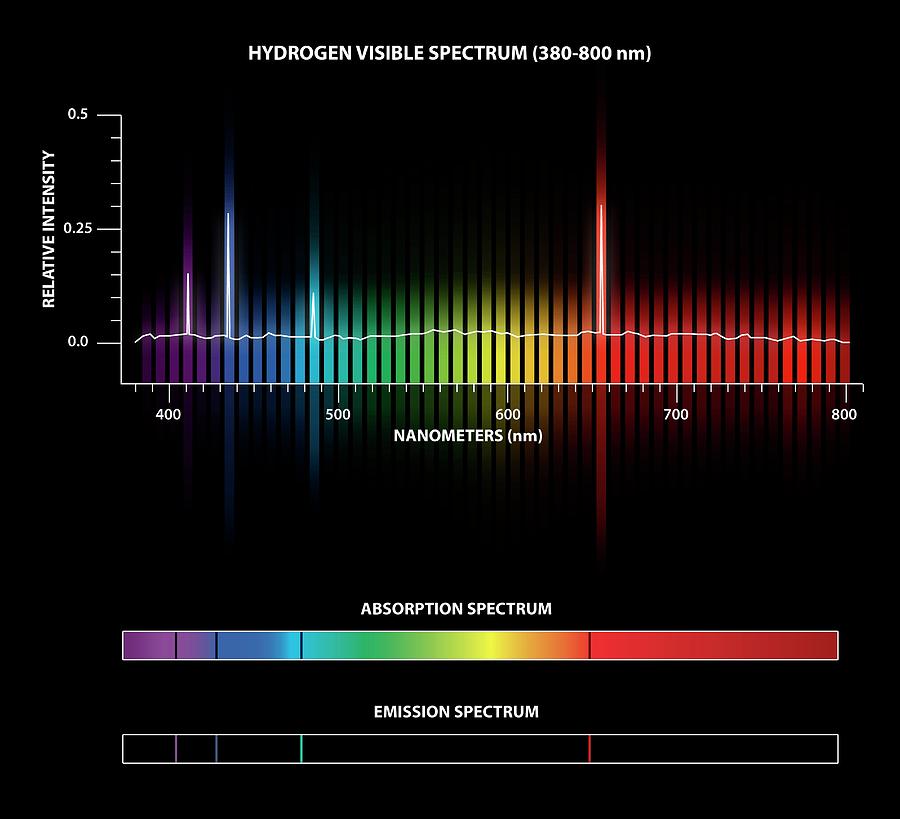
 Structure 1.3.6-In an emission spectrum, the limit of convergence at higher frequency corresponds to ionization.Structure 1.3.5-Each orbital has a defined energy state for a given electron configuration and chemical environment, and can hold two electrons of opposite spin.Structure 1.3.4-A more detailed model of the atom describes the division of the main energy level into s, p, d and f sublevels of successively higher energies.Structure 1.3.3-The main energy level is given an integer number, n, and can hold a maximum of2n^2 electrons.Structure 1.3.2-The line emission spectrum of hydrogen provides evidence for the existence of electrons in discrete energy levels, which converge at higher energies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
